RapR / UniRapR / RapR TAP
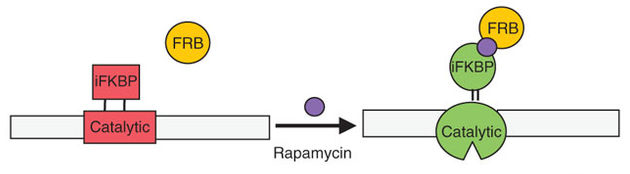
Schematic representation of the approach used to regulate the catalytic activity of kinases. A fragment of FKBP is inserted at a position in the catalytic domain where it allosterically inhibits catalytic activity. Binding to rapamycin and FRB restores activity.
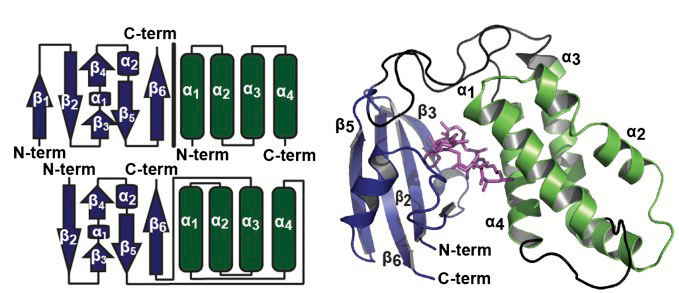
UniRapR (blue, subdomain-A; green, subdomain-B) protein was built by fusing FKBP and FRB. Using uniRapR there is no need to express two constructs. The fused FRB and FKBP, when inserted in the target protein, respond to rapamycin and activate it. This approach has been extended to apply to molecules other than kinases. See the "loopology" approach page.
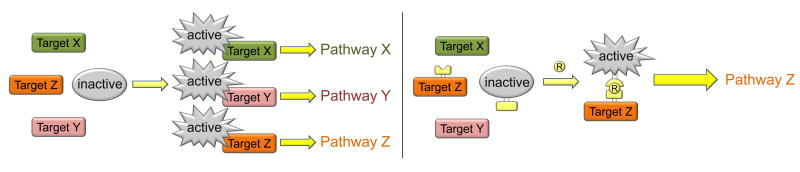
Unlike with normal kinases that interact with multiple substrates, with RapR-TAP a specific kinase is activated to interact with only one specific target (right). Activation occurs when addition of rapamycin leads to interaction of two engineered domains (yellow), on the kinase and on the target.
References
RapR
Chu, P-H., Tsygankov, D., Berginski, M.E., Dagliyan, O., Gomez, S.M., Elston, T.C., Karginov, A.V. and Hahn, K.M. Engineered kinase activation reveals unique morphodynamic phenotypes and associated trafficking for Src family isoforms. Proc. Natl. Acad. Sci. U.S.A.,111(34): 12420–12425, 2014. PMC4151743. Online article | Free PMC article
Karginov, A.V., Ding, F., Kota, P., Dokholyan, N.V., and Hahn, K.M. Engineered allosteric activation of kinases in living cells. Nature Biotech, July; 28(7):743-7, 2010. PMC2902629. Online article | Free PMC article
UniRapR
Dagliyan, O., Karginov, A.V., Yagishita, S., Gale, M.E., Wang, H., DerMardirossian, C., Wells, C.M., Dokholyan, N.V., Kasai, H. and Hahn, K.M. Engineering Pak1 Allosteric Switches. ACS Synth. Biol., 6 (7): 1257–1262, 2017. PMC5562282. Online article | Free PMC article
Dagliyan, O., Tarnawski, M., Chu, P-H., Shirvanyants, D., Schlichting, I., *Dokholyan, N.V., and *Hahn, K.M. Engineering extrinsic disorder to control protein activity in living cells. Science, 354(6318): 1441-1444, 2016. PMC5362825. View free in Science online: Full text & PDF | Free PMC article
Dagliyan, O., Shirvanyants, D., Karginov, A.V., Ding, F., Fee, L., Chandrasekaran, S.N., Freisinger, C.M., Smolen, G.A., Huttenlocher, A., *Hahn, K.M., *Dokholyan, N.V. Rational design of a ligand-controlled protein conformational switch. Proc. Natl. Acad. Sci. U.S.A., 110(17):6800-4, 2013. PMC3637791. Online article | Free PMC article
RapR TAP
Karginov, A., Tsygankov, D., Berginski, M., Chu, P-H., Trudeau, E., Yi, J.J., Gomez, Shawn, Elston, T.C. and Hahn, K.M. Dissecting motility signaling through activation of specific Src-effector complexes. Nature Chem. Bio., 10(4): 286-90, 2014. PMC4064790. Online article | Free PMC article
Methods articles
Dagliyan, O., Dokholyan, N.V., and Hahn, K.M. Engineering proteins for allosteric control by light or ligands. Nature Protocols, 14(6): 1863-1883, 2019. PMC6648709. Online article | Free PMC article
Karginov, A.V. and Hahn, K.M. Allosteric activation of kinases: design and application of RapR kinases. Curr. Protoc. Cell Biol., 53: 14.13.1 – 14.13.16, 2011. PMC3269071. Online article | Free PMC article
~ Updated 04/09/2021